Most people who had this type of reaction had it within minutes of getting the vaccine and responded to treatment epinephrine. Clinical trials patients are usually faceless.
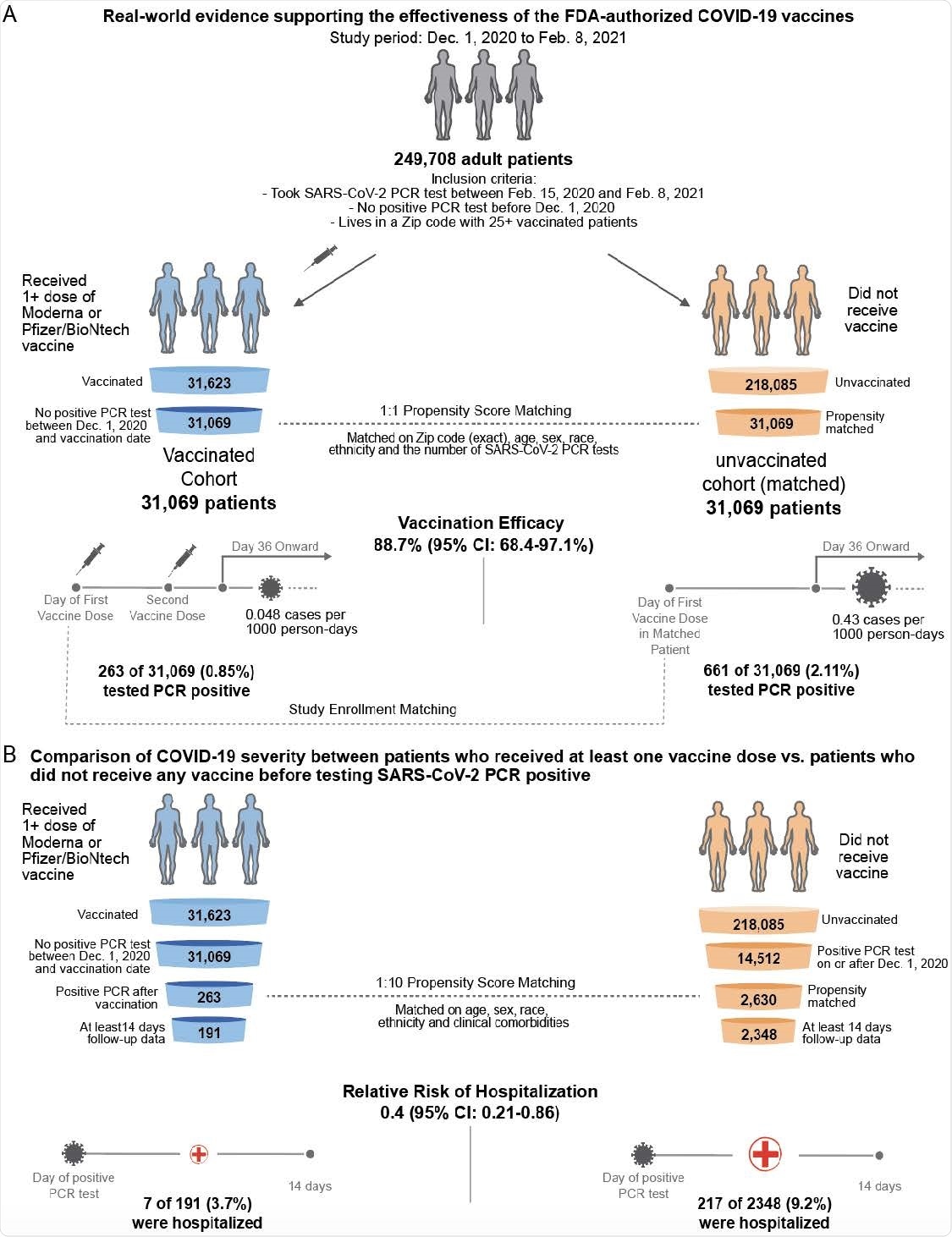
Study Shows Real World Effectiveness Of Moderna And Pfizer Biontech Vaccines
A review Patient Saf Surg.

Moderna vaccine reviews from patients. FDA may issue an EUA when certain criteria are met which. These vaccines are based on the mRNA vaccine platform and were developed by PfizerBioNTech and Moderna. At best vaccine takers may experience just a hint of arm pain.
Overall across all countries where trials were conducted the Johnson Johnson vaccine showed efficacy of 67 at preventing moderate to severecritical COVID-19 infection at least 14 days after. More side effects were reported among patients who received the Moderna vaccine rather than the Pfizer shot. Patients with inflammatory bowel diseases IBD do not appear to have increased risk of side effects from the Pfizer or Moderna COVID-19 vaccines according to a recent Cedars-Sinai study.
In the patient fact sheet for the Moderna vaccine the FDA notes that symptoms of myocarditis or pericarditis tend to appear within a few days following receipt of the second dose. About 73 of Moderna patients had pain at the injection site compared to 65 of Pfizer patients. The EUA letters for both the Moderna and the PfizerBioNTech vaccines contain provisions obliging post-authorization observational studies and label pregnant women as a population of interest for these studies citing a lack of data regarding vaccine risk in pregnancy.
Now doctors at Massachusetts General Hospital are reporting an alarming -- though ultimately harmless -- reaction in an increasing number of patients after initial doses of the Cambridge biotech companys product. The Moderna vaccine was 941 effective at preventing symptomatic COVID-19 starting from 14 days after the second dose was administered. 2 can feel like a full-blown flu with nausea vomiting and body aches.
The Moderna vaccine is for use only in adults. The Moderna vaccine has been shown to have an efficacy of approximately 941 per cent in protecting against COVID-19 starting 14 days after the first dose. The two vaccines are very similar but they have a few key differences that make Modernas vaccine more flexible as US Health and Human.
Does it work against new variants. A detailed description of the study which is not yet peer-reviewed. Moderna vaccine still effective after six months.
And 51 of Moderna patients said they had chills fatigue and body side effects where as 48 of Pfizer patients. Adverse effects on fetal. At worst the day after shot No.
The side effects tend to be benign with. COVID-19 vaccine is experimental and all of its risks are not yet known. This vaccine may help your body develop immunity to SARS-CoV-2.
BOSTON Modernas Covid-19 vaccine has already achieved a degree of notoriety for uncomfortable side effects following the booster dose. According to the study Moderna patients had more reactions. However this vaccine has not been approved to prevent or treat coronavirus or COVID-19.
How effective is the Johnson Johnson vaccine. Moderna Covid-19 VaccineIntramuscular received an overall rating of 2 out of 10 stars from 2 reviews. The Moderna COVID-19 Vaccine is a vaccine and may prevent you from getting COVID-19 the virus that causes fever or chills.
The Moderna vaccine fact sheet specifically alludes to a reproductive toxicity study in female rats. We dont yet have similar data on the Moderna vaccine. Trial volunteer Ian Haydon.
The findings reveal that the Moderna vaccine induces higher and faster antibody responses than the PfizerBioNTech vaccine. The COVID-19 vaccine does not contain coronavirus and cannot give you COVID-19. How Well the Vaccine Works.
Based on evidence from clinical trials in people aged 18 years and older the Moderna vaccine was 941 effective at preventing laboratory-confirmed COVID-19 infection in people who received two doses and had no evidence of being previously infected. Review the safety of Covid-19 mRNA vaccines. See what others have said about Moderna Covid-19 VaccineIntramuscular.
Published safety and efficacy trials reported high efficacy rates of 94-95 after two interval doses in conjunction with limited side effects and a low rate of. The Moderna COVID-19 Vaccine has not undergone the same type of review as an FDA- approved or cleared product. With Moderns Covid-19 vaccine candidate it has a much more visible advocate.
In all six cases under review the patients developed symptoms one to three weeks after receiving the vaccine. Based on the evidence so far the new variants of SARS-CoV-2 including the B117 and the 501YV2 do not alter the effectiveness of the Moderna mRNA.

Covid 19 Vaccine Frequently Asked Questions City Of Hamilton Ontario Canada

Jj Vaccine Single Shot Johnson Johnson Covid Vaccine 66 Percent Effective Against Moderate And Severe Illness The Washington Post

Covid 19 Vaccines What You Need To Know Seattle Cancer Care Alliance

Vaccine Side Effects Enlarged Heart Warning For Pfizer Moderna Jabs In Usa Hindustan Times
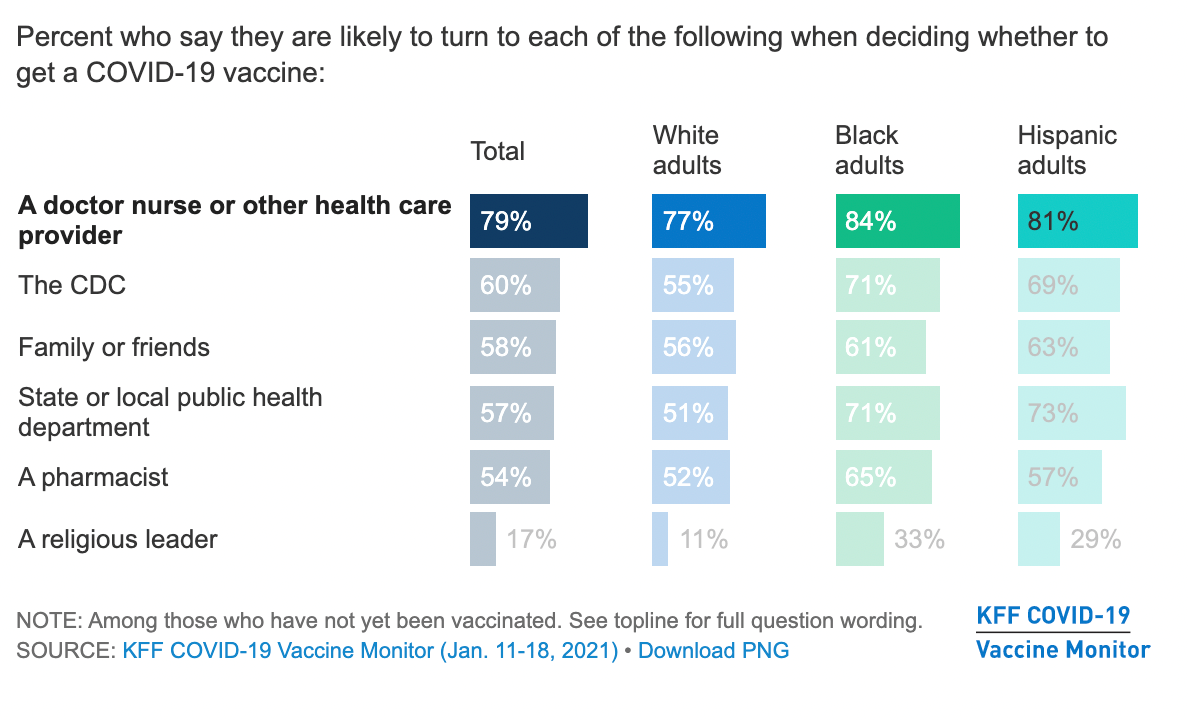
Covid 19 Vaccine Hesitancy Addressing Common Concerns Beaumont Health

Covid Vaccine Infographic Curehht
Covid 19 Risks And Side Effects Of Vaccination Science In Depth Reporting On Science And Technology Dw 20 01 2021
Johnson Johnson Vaccine Rolls Out Amid Concerns About Bias From Bad Publicity Npr

Moderna Edges Pfizer In Covid 19 Vaccine Effectiveness And Refrigeration
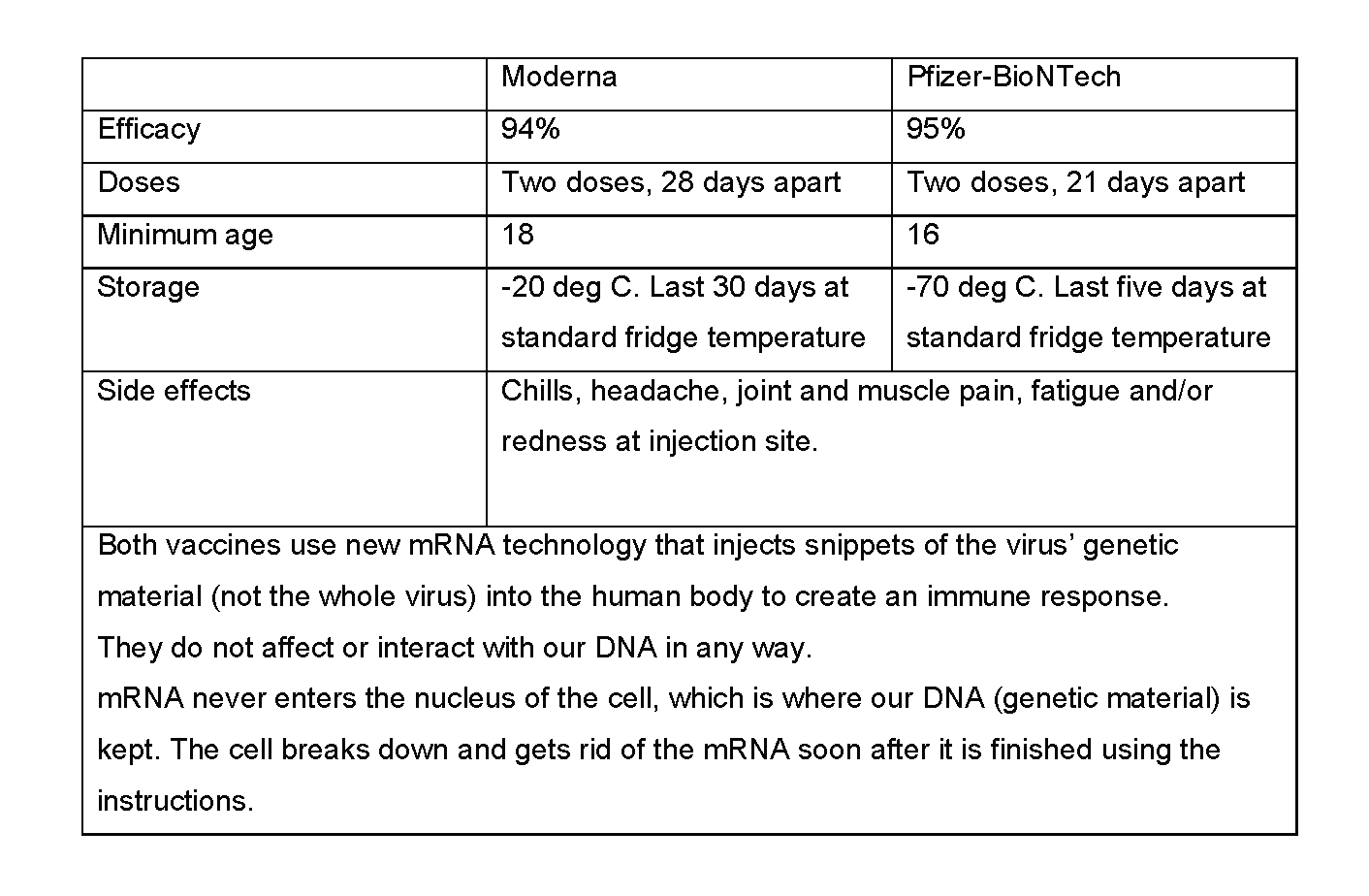
Pfizer Vs Moderna Covid 19 Vaccines International Medical Clinic Imc
A One Shot Covid 19 Vaccine Johns Hopkins Medicine

Covid 19 Vaccine News Faq Advocate Aurora Health
Fda Adds Warning Of Rare Heart Inflammation To Pfizer Moderna Vaccines

Us Begins Study On Covid 19 Vaccine Side Effects Pfize And Moderna Vaccines Reactions Wion News Youtube

My Experience With The Moderna Vaccine What You Can Expect Wbtw

Covid 19 Vaccination Updates Nuhs National University Health System